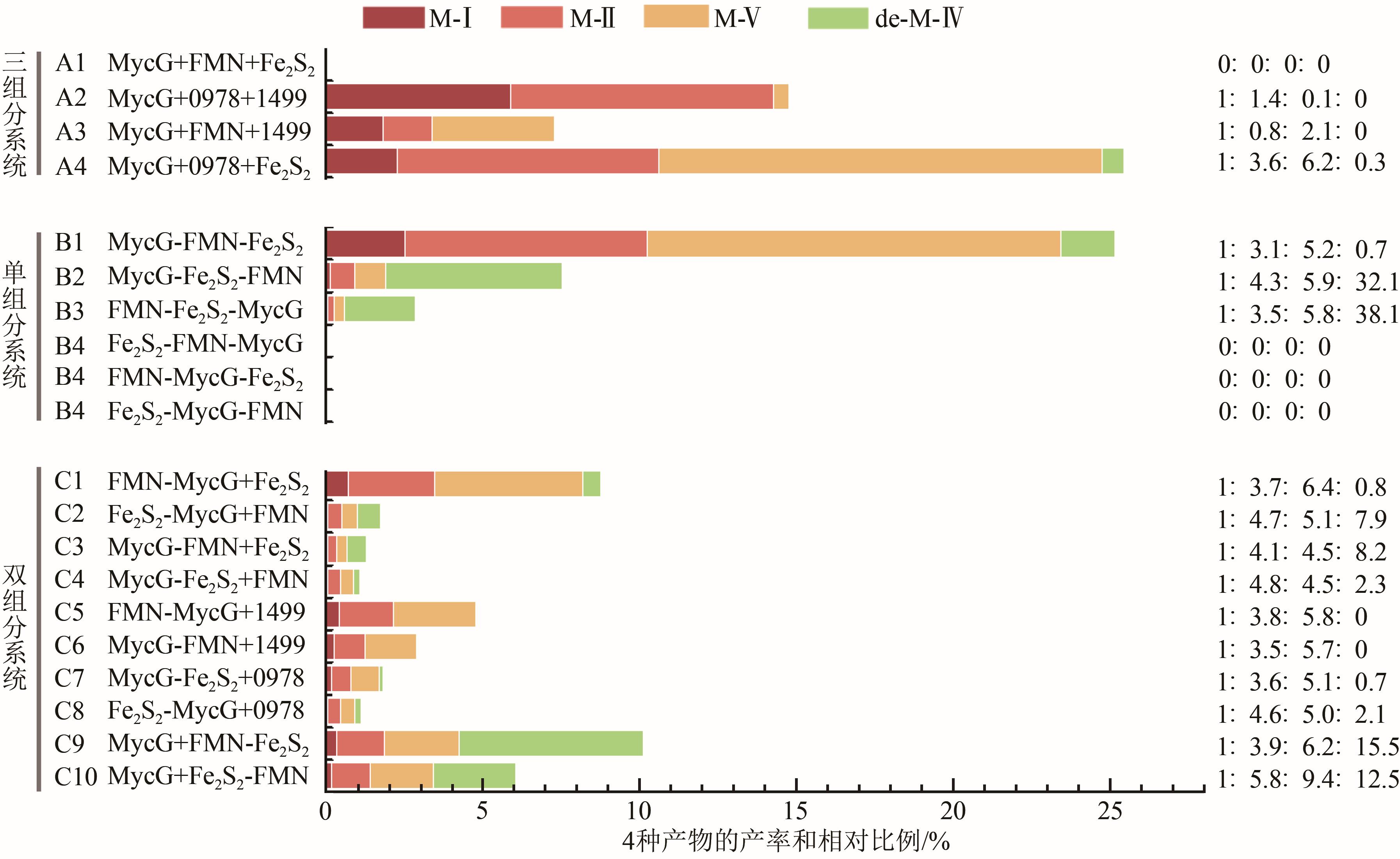
| 1 |
GIRVAN H M, MUNRO A W. Applications of microbial cytochrome P450 enzymes in biotechnology and synthetic biology[J]. Current Opinion in Chemical Biology, 2016, 31: 136-145.
|
| 2 |
GUENGERICH F P, MUNRO A W. Unusual cytochrome P450 enzymes and reactions[J]. Journal of Biological Chemistry, 2013, 288(24): 17065-17073.
|
| 3 |
LAMB D C, WATERMAN M R. Unusual properties of the cytochrome P450 superfamily[J]. Philosophical Transactions of the Royal Society of London Series B, Biological Sciences, 2013, 368(1612): 20120434.
|
| 4 |
GUENGERICH F P. Common and uncommon cytochrome P450 reactions related to metabolism and chemical toxicity[J]. Chemical Research in Toxicology, 2001, 14(6): 611-650.
|
| 5 |
SEVRIOUKOVA I F, GARCIA C, LI H Y, et al. Crystal structure of putidaredoxin, the [2Fe-2S] component of the P450cam monooxygenase system from Pseudomonas putida [J]. Journal of Molecular Biology, 2003, 333(2): 377-392.
|
| 6 |
KEIZERS P H J, MERSINLI B, REINLE W, et al. A solution model of the complex formed by adrenodoxin and adrenodoxin reductase determined by paramagnetic NMR spectroscopy[J]. Biochemistry, 2010, 49(32): 6846-6855.
|
| 7 |
MATSUBARA H, SASAKI R M, CHAIN R K. Spinach ferredoxin (Ⅰ): Amino acid composition and terminal sequences[J]. Journal of Biological Chemistry, 1968, 243(8): 1725-1731.
|
| 8 |
RUETTINGER R T, FULCO A J. Epoxidation of unsaturated fatty acids by a soluble cytochrome P450-dependent system from Bacillus megaterium [J]. Journal of Biological Chemistry, 1981, 256(11): 5728-5734.
|
| 9 |
JACKSON C J, LAMB D C, MARCZYLO T H, et al. A novel sterol 14α-demethylase/ferredoxin fusion protein (MCCYP51FX) from Methylococcus capsulatus represents a new class of the cytochrome P450 superfamily[J]. Journal of Biological Chemistry, 2002, 277(49): 46959-46965.
|
| 10 |
JACKSON R G, RYLOTT E L, FOURNIER D, et al. Exploring the biochemical properties and remediation applications of the unusual explosive-degrading P450 system XplA/B[J]. Proceedings of the National Academy of Sciences of the United States of America, 2007, 104(43): 16822-16827.
|
| 11 |
MUNRO A W, DAFF S, COGGINS J R, et al. Probing electron transfer in flavocytochrome P-450 BM3 and its component domains[J]. European Journal of Biochemistry, 1996, 239(2): 403-409.
|
| 12 |
MCLEAN K J, LUCIAKOVA D, BELCHER J, et al. Biological diversity of cytochrome P450 redox partner systems[J]. Advances in Experimental Medicine and Biology, 2015, 851: 299-317.
|
| 13 |
LI S Y, PODUST L M, SHERMAN D H. Engineering and analysis of a self-sufficient biosynthetic cytochrome P450 PikC fused to the RhFRED reductase domain[J]. Journal of the American Chemical Society, 2007, 129(43): 12940-12941.
|
| 14 |
NODATE M, KUBOTA M, MISAWA N. Functional expression system for cytochrome P450 genes using the reductase domain of self-sufficient P450RhF from Rhodococcus sp. NCIMB 9784[J]. Applied Microbiology and Biotechnology, 2006, 71(4): 455-462.
|
| 15 |
MCLEAN K J, HANS M, MEIJRINK B, et al. Single-step fermentative production of the cholesterol-lowering drug pravastatin via reprogramming of Penicillium chrysogenum [J]. Proceedings of the National Academy of Sciences of the United States of America, 2015, 112(9): 2847-2852.
|
| 16 |
ANZAI Y, LI S Y, CHAULAGAIN M R, et al. Functional analysis of MycCI and MycG, cytochrome P450 enzymes involved in biosynthesis of mycinamicin macrolide antibiotics[J]. Chemistry & Biology, 2008, 15(9): 950-959.
|
| 17 |
ZHANG W, LIU Y, YAN J Y, et al. New reactions and products resulting from alternative interactions between the P450 enzyme and redox partners[J]. Journal of the American Chemical Society, 2014, 136(9): 3640-3646.
|
| 18 |
GUENGERICH F P, MARTIN M V, SOHL C D, et al. Measurement of cytochrome P450 and NADPH-Cytochrome P450 reductase[J]. Nature Protocols, 2009, 4(9): 1245-1251.
|
| 19 |
ZHANG W, DU L, LI F W, et al. Mechanistic insights into interactions between bacterial class I P450 enzymes and redox partners[J]. ACS Catalysis, 2018, 8(11): 9992-10003.
|
| 20 |
TASNIM H, LANDRY A P, FONTENOT C R, et al. Exploring the FMN binding site in the mitochondrial outer membrane protein mitoNEET[J]. Free Radical Biology and Medicine, 2020, 156: 11-19.
|
| 21 |
HEIKAL A A. Intracellular coenzymes as natural biomarkers for metabolic activities and mitochondrial anomalies[J]. Biomarkers in Medicine, 2010, 4(2): 241-263.
|
| 22 |
LACOUR T, OHKAWA H. Engineering and biochemical characterization of the rat microsomal cytochrome P4501A1 fused to ferredoxin and ferredoxin-NADP+ reductase from plant chloroplasts[J]. Biochimica et Biophysica Acta-Protein Structure and Molecular Enzymology, 1999, 1433(1/2): 87-102.
|
| 23 |
WANG Q, HUANG X N, ZHANG J J, et al. Engineering self-sufficient aldehyde deformylating oxygenases fused to alternative electron transfer systems for efficient conversion of aldehydes into alkanes[J]. Chemical Communications, 2014, 50(33): 4299.
|
| 24 |
HUNTER D J B, ROBERTS G A, OST T W B, et al. Analysis of the domain properties of the novel cytochrome P450 RhF[J]. FEBS Letters, 2005, 579(10): 2215-2220.
|
| 25 |
TRIPATHI S, LI H Y, POULOS T L. Structural basis for effector control and redox partner recognition in cytochrome P450[J]. Science, 2013, 340(6137): 1227-1230.
|
| 26 |
ZHANG L L, XIE Z Z, LIU Z W, et al. Structural insight into the electron transfer pathway of a self-sufficient P450 monooxygenase[J]. Nature Communications, 2020, 11: 2676.
|
| 27 |
SADEGHI S J, MEHARENNA Y T, FANTUZZI A, et al. Engineering artificial redox chains by molecular 'LEGO'[J]. Faraday Discussions, 2000(116): 135-153.
|
| 28 |
LI Z, JIANG Y Y, GUENGERICH F P, et al. Engineering cytochrome P450 enzyme systems for biomedical and biotechnological applications[J]. Journal of Biological Chemistry, 2020, 295(3): 833-849.
|